
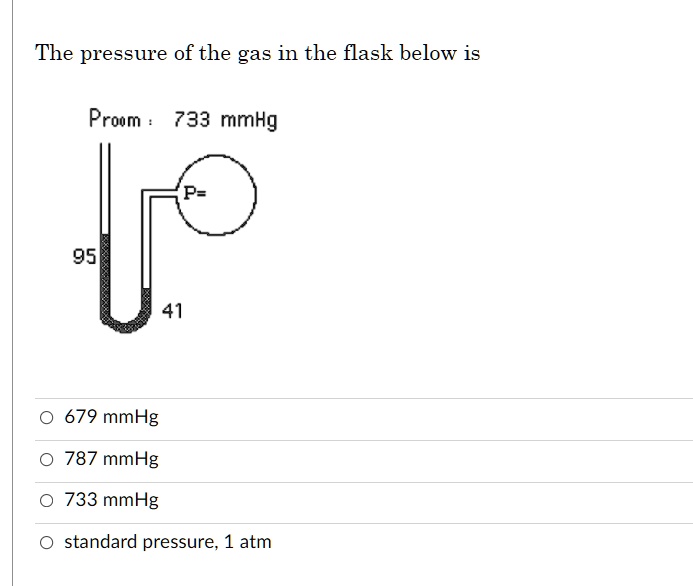
Calculate how much of this gravel is required to attain a specific depth in a cylindrical, quarter cylindrical or in a rectangular shaped aquarium or pond List of these foods starting with the highest contents of Tocotrienol, gamma and the lowest contents of Tocotrienol, gamma Gravels, Substances and OilsĬaribSea, Marine, CORALine, Volcano Beach weighs 865 kg/m³ (54.00019 lb/ft³) with specific gravity of 0.865 relative to pure water. How much of a stickler there on that? Um, I'm gonna leave it at 81.7 millimeters of mercury.Millimeters of mercury to newtons per square meter conversion cardsĭECORATING ICING BLUE, UPC: 071169730022 weigh(s) 304 grams per metric cup or 10.2 ounces per US cup, and contain(s) 417 calories per 100 grams (≈3.53 ounces) ĥ50 foods that contain Tocotrienol, gamma. You can ask your teacher what they prefer. Ah, so it's it would be in my brain acceptable to use three sig figs here as your smallest. P PV Solution: Mass (g) of gas mass of bulb + gas mass of bulb 82.786 82.561 0.225 g T 22C + 273.15 295 K P (733 mmHg) 1 atm 760 mmHg V. However, my brain sees this like Okay, we have the same precision in both of our temperatures. Technically, our smallest number of sig figs is, too. But doing so correctly will give you a pressure of 81 0.7 millimeters of mercury. You might want to do it in multiple steps to make sure using proper parentheses. Be very careful when you're plugging this into your calculator. And so our answer is gonna have the units of millimeters of mercury, which is a unit of pressure, which is what we want. Uh, would you look at that? Are middle leaders Cancel our Kelvin's cancel. So we will also multiply this side by 232.

Whatever we do to one side of the equation, we must do to the other. But whatever we do to one side, if we multiply this here Oh, look at that.
733 mmhg to atm plus#
By the reciprocal of these boys, this will be 252 mil leaders on the bottom and ah, zero plus 2 73 purple. We just need to revisit Our algebra will multiply both sides. So we convert to Kelvin when we're making use of the gas laws from here to isolate our pressure and find our answer. You don't want to have to answer that question for your gas problem. How maney nothing's Can you fit into something. Expanded volume 252 milliliters divided by our new pressure 0.0 Excuse degree Celsius, which we need to convert. Converted Delvin By adding to 73 we set this equal to our mystery pressure. 165 millimeters of mercury Times are a volume of 135 mil leaders All this divided by our temperature, which is 22.5. Choose a vacuum pressure in millimeters of mercury column from 0.1 to 1000 mmHg to cross reference the converted pressure in millimeters of water column. You can look at the video for problem number eight. 0.96842 Atmospheres (atm) Visit 736 Atmospheres to mmHg Conversion Millimeter Mercury (0C) : Millimeter of mercury is a small pressure unit which represents the pressure pushing down due to gravity of any volume of liquid mercury which is 1mm high. If you'd like to see the derivation of our combined gas law. Make use of the combined gas law pressure times volume divided by temperature of our first situation equal to the pressure, volume and temperature of our second situation in Koven will come to why in just a moment let's not dilly dally any time and just plug our measurements straight in there. We're gonna once again just like problem. Ah, and we've been tasked to find What is the new pressure in this situation? Find this answer.
Who it is cooled on down zero degrees Celsius. Oh, is it not in Kelvin? And that's exerting a pressure of 165 millimeters of mercury on the walls are flask and we're tasked with transferring this to a new flask with different conditions. We've got a volume of 135 milliliters at 22 a half degrees Celsius. They keep doing that to us, but it's okay. We're going to take a stab at Problem number 12 from chapter 12 where we've been once again handed a flask of gas.
